Research in the Braunschweig Group
Research in the group explores the interface of organic chemistry with biology and materials science to find new solutions to energy, health, and environmental challenges. Supramolecular interactions, nanomaterials, and molecular printing are employed to create functional hierarchical structures from relatively simple starting materials. Emphasis is placed on the rational design of target molecules and a fundamental understanding of their assembly and function.
See our publications.
Supramolecular Materials
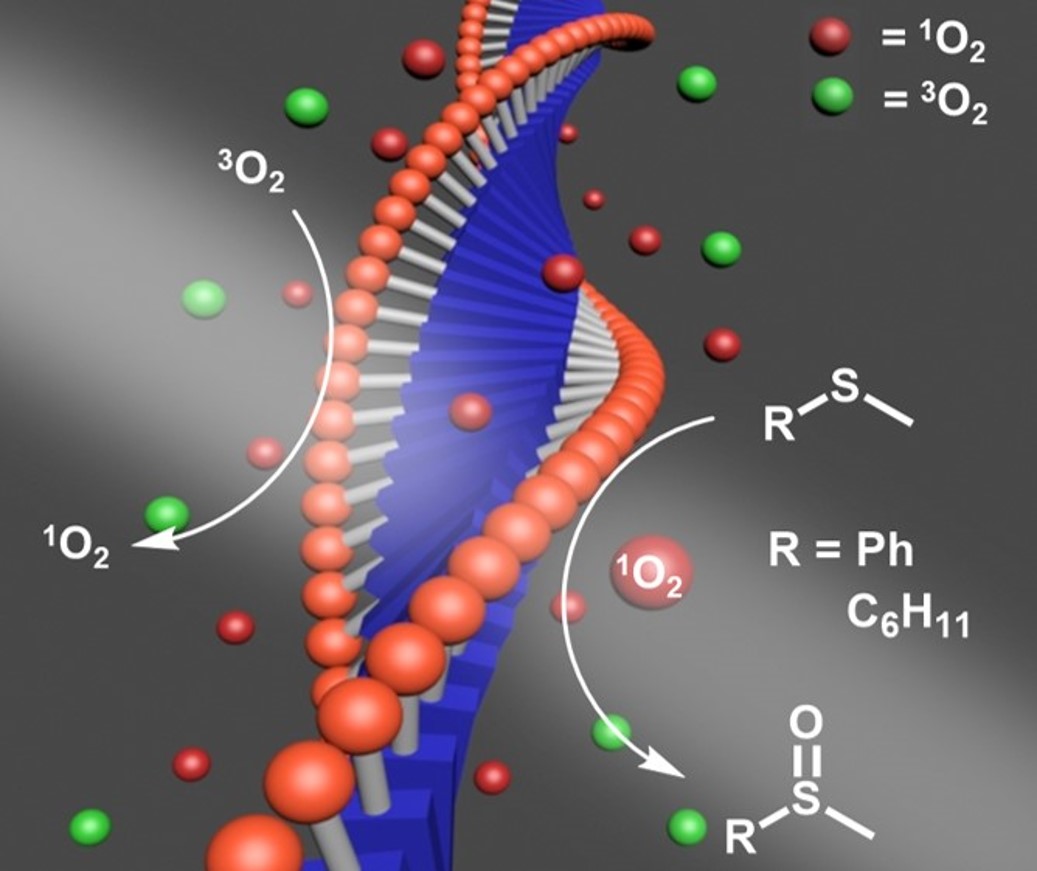
Using organic synthesis, our lab designs new materials with self-assembling behavior for photophysical applications including energy harvesting and catalysis.
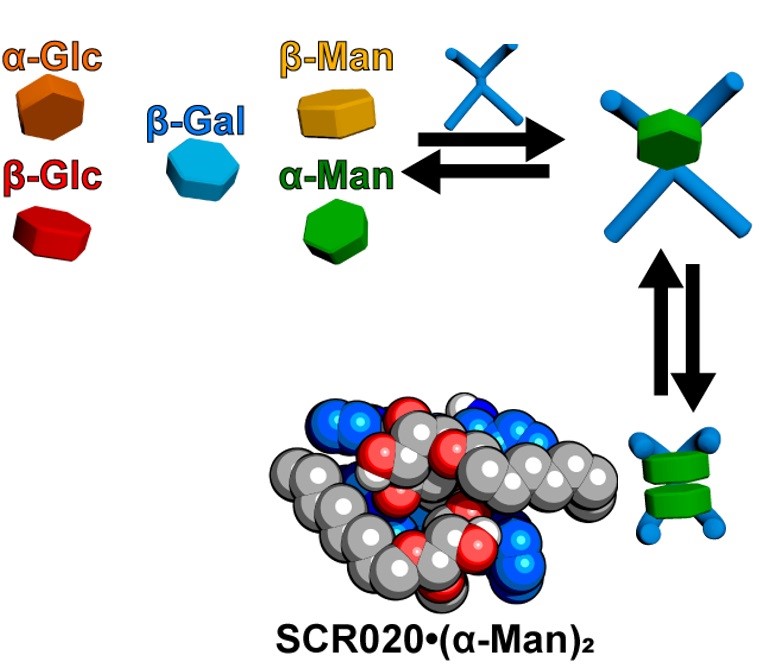
Synthetic Carbohydrate Receptors
There is a pressing need for safe and effective antivirals, which we aim to meet by designing molecules that bind sugars with tailorable specificity.
Molecular Printing and Nanolithography
Rapid advancement of surface and microarray science are limited by the lack of available tools, thus by developing new molecular printing technologies, high-throughput screening of surface chemistries and studies are made possible.

Comparative Mucomics
We utlize a novel comparative approach modeled after modern omics-style analysis to study the chemical structures and physical properties of mucus, one of the most important and diverse biological materials.

Biomimetic Glycopolymers
Glycoproteins are used throughout nature to meet numerous material needs and our lab seeks to replicate and improve upon these molecules using a combination of carbohydrate and polymer chemistry.

Mechanochemistry
Mechanical force is an invaluable tool to drive chemical reactions, but has not achieved mainstream use. Our lab seeks to apply new approaches of to expand the applications and understanding of mechanochemical processes to rival those using light, heat, and charge.
